About ADMELOG® (insulin lispro) injection 100 Units/mL
ADMELOG is a fast-acting mealtime insulin for adults with type 1 or type 2 diabetes, and for children (3 years and older) with type 1 diabetes, that:
- Is proven to lower A1C
- Helps control blood sugar spikes that happen naturally after you eat
You may be able to save if you don’t have prescription insurance for your mealtime insulin.
Find out more with ADMELOG FAQs

ADMELOG is available in the ADMELOG SoloStar® pen or a vial, and can be used in a pump.†
†See your insulin infusion pump system’s instructions for use to see if ADMELOG can be used with your pump system.
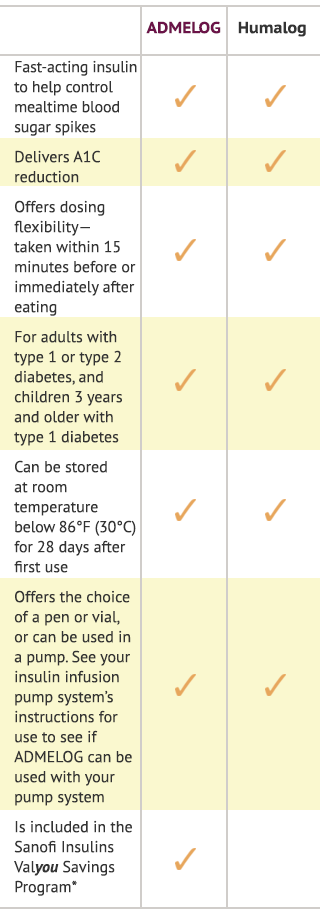
ADMELOG vs Humalog®
See how ADMELOG compares with Humalog, one of the most prescribed mealtime insulins. Any change in your medication should be discussed with your doctor.
If you’re uninsured or insured and prefer to pay cash outside of your prescription drug benefit, we have the Insulins Valyou Savings Program* for you! Learn more.
Frequently Asked Questions
If you and your doctor are considering ADMELOG, having the right information can help.
Click on the questions below to get answers to frequently asked questions.
Side Effects 
- Low blood sugar (hypoglycemia).
- Allergic reactions, including reactions at the injection site, skin thickening or pits at the injection site (lipodystrophy), itching, and rash.
- Low blood sugar (hypoglycemia).
- Serious allergic reactions (whole body reaction).
- Low potassium in your blood (hypokalemia).
- Heart failure.
- Any of these signs or symptoms of a severe allergic reaction: a rash over your whole body, trouble breathing, a fast heartbeat, feeling faint, sweating, swelling of your face, tongue, or throat.
- Extreme drowsiness, dizziness, or confusion.
The most common side effects of ADMELOG include:
These are not all the possible side effects of ADMELOG. It is important to review the labeling that comes with your ADMELOG prescription and speak to your doctor about all possible side effects. Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
ADMELOG may cause serious side effects that can lead to death, including:
Get emergency medical help right away if you have:
Hypoglycemia is the most common adverse event associated with insulin-containing therapies.
About ADMELOG 
- Mealtime insulins containing insulin lispro
- Used to help control blood sugar spikes that occur when you eat
- Taken within 15 minutes before or immediately after a meal
ADMELOG is a fast-acting mealtime insulin that helps to control blood sugar. It is used to attempt to mimic your body’s natural insulin response to blood sugar spikes after meals. It is to be taken within 15 minutes before meals or immediately after.
ADMELOG contains insulin lispro (100 Units/mL), a man-made insulin.
Long-acting insulin (along with diet and exercise) may not be enough to control your blood sugar spikes that happen when you eat. That may be why your doctor has recommended adding ADMELOG.
Unlike long-acting insulin you take once a day, ADMELOG is a fast-acting insulin you take at mealtimes to help you manage blood sugar spikes that happen after you eat. It can help to lower your A1C.
ADMELOG is not a generic for Humalog. However, both ADMELOG and Humalog are:
Be sure to review the possible side effects with your doctor. Your doctor will determine what is right for you.
If your doctor is recommending ADMELOG, you should know that it is a mealtime insulin used to help control blood sugar spikes that occur when you eat.
Your doctor may need to adjust your mealtime insulin dose if you are being switched from NovoLog to ADMELOG.
The cost of mealtime insulin depends on many factors including your health insurance, your dose, or if you are using a pump, pen, or vial.
If you’re uninsured or insured and prefer to pay cash outside of your prescription drug benefit, we have the Insulins Valyou Savings Program* for you.
You will pay $35* per 30-day supply of any one or combination of Sanofi Insulins. Eligibility restrictions apply. Patients must fill all their Sanofi Insulin prescriptions at the same time, together each month. Learn more.
Insulins Valyou Savings Program: This savings program is not insurance. For a complete list of participating brands, products, and National Drug Codes (NDCs) Click Here. This offer is only valid for those who are uninsured or those who are insured by a prescription plan but are not using such insurance and will be paying the full retail price for the medication. Void where prohibited by law. The Savings Program applies to the cost of medication. There are other relevant costs associated with overall treatment. You may not submit claims for reimbursement to any third-party payor, including any government healthcare plan (e.g., Medicare, Medicaid, DOD, VA, TRICARE) or similar federal or state programs for Sanofi Insulin prescriptions when using this Program. You may not seek to have your out-of-pocket costs or the full retail price of the Sanofi Insulin count toward your deductible, true-out-of-pocket (TrOOP), maximum out-of-pocket (MOOP), or any other out-of-pocket caps associated with any insurance coverage. Sanofi reserves the right to rescind, revoke, terminate, or amend this offer, eligibility, and terms of use at any time without notice. Upon registration, patients will receive all program details. For questions regarding your eligibility or benefits, or if you wish to discontinue your participation, call the Insulins Valyou Savings Program at (833) 813-0190 (8:00 am-8:00 pm EST, Monday-Friday).
Using ADMELOG 
-
You should only use your pen for up to 28 days after its first use. Throw away the ADMELOG SoloStar pen you are using after the 28th day, even if it still has insulin left in it.
Be sure to put used needles and syringes in an FDA-cleared disposal container right away after use.
-
While using ADMELOG, do not:
- Drive or operate heavy machinery, until you know how ADMELOG affects you.
- Drink alcohol or use prescription or over-the-counter medicines that contain alcohol.
-
Before first use
- Keep new pens in the refrigerator between 36°F and 46°F (2°C and 8°C) until the expiration date. Discard if expired.
- Do not freeze. Do not use ADMELOG if it has been frozen.
-
After first use
- Keep your pen at room temperature below 86°F (30°C).
- Must be used within 28 days after first use.
- Keep your pen away from heat or light.
- Store your pen with the pen cap on.
- Do not put your pen back in the refrigerator.
- Do not store your pen with the needle attached.
- Keep out of the reach of children.
-
Your dose of ADMELOG may need to change because of:
- A change in physical activity or exercise.
- Changes in meal patterns.
- Weight gain or loss.
- Increased stress.
- Illness.
- A change in diet.
- Changes in kidney or liver functions.
- Other medicines you take.
-
You can inject ADMELOG in four areas of your body:
- Anywhere in your stomach area, except for a two-inch radius around your navel
- In the fatty tissue on the outer back area of your upper arm
-
Rotate your injection sites with each dose to reduce your risk of getting lipodystrophy (pitted or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites. Do not use the same spot for each injection or inject where the skin is pitted, thickened, lumpy, tender, bruised, scaly, hard, scarred or damaged.
- In your thigh, away from your knee
- In your buttocks
-
You should take ADMELOG within 15 minutes before or immediately after eating a meal.
-
You can take ADMELOG using:
- A SoloStar disposable prefilled injection pen.
- A syringe and vial.
- An insulin pump. See your insulin infusion pump system’s instructions for use to see if ADMELOG can be used with your pump system.
-
Speak to your doctor about what option may be appropriate for you.
- If you use the SoloStar pen or the vial, read the detailed Instructions for Use that come with your ADMELOG. If you use a pump, see your insulin infusion pump system’s instructions for use.
- Speak with your healthcare provider about how to use ADMELOG, and use it exactly as directed.
- Know the amount of ADMELOG you use. Do not change the amount of ADMELOG you use unless your healthcare provider tells you to. The amount of insulin and the best time for you to take your insulin may need to change if you take a different type of insulin.
- Check your insulin label each time you inject to make sure you are using the correct insulin.
- Do not reuse needles. Using a new needle for each injection lowers your risk of getting an infection or getting the wrong dose of ADMELOG due to a blocked needle.
- Keep out of reach of children.
- Have liver or kidney problems
- Take other medicines, especially ones commonly called thiazolidinediones (TZDs)
- Have heart failure or other heart problems. If you have heart failure, it may get worse while you take TZDs with ADMELOG
- Are pregnant or planning to become pregnant, or are breastfeeding. It is not known if ADMELOG may harm your unborn or breastfeeding baby
- Prescription medicines.
- Over-the-counter medicines.
- Vitamins.
- Herbal supplements.
- Shortness of breath.
- Swelling of your ankles or feet.
- Sudden weight gain.
- Have low blood sugar (hypoglycemia).
- Have an allergy to ADMELOG or any of the ingredients in ADMELOG.
Be sure to tell your healthcare provider about all of your medical conditions, including if you:
Tell your healthcare provider about all of the medicines you take, including:
Before you start using ADMELOG, talk to your healthcare provider about low blood sugar and how to manage it.
You should ask your doctor which medicines (including over-the-counter and other prescription medicines, vitamins, and herbal supplements) are safe to take if you are using ADMELOG.
Taking certain diabetes pills called thiazolidinediones (TZDs) with ADMELOG may cause heart failure in some people. This can happen even if you have never had heart failure or heart problems before. If you have already had heart failure, it may get worse while you take TZDs with ADMELOG. Your healthcare provider should monitor you closely while you are taking TZDs with ADMELOG.
Tell your healthcare provider if you have any new or worse symptoms of heart failure, including:
Treatment with TZDs and ADMELOG may need to be adjusted or stopped by your healthcare provider if you have new or worse heart failure.
Do not use ADMELOG if you:
- Active ingredient: insulin lispro
- Inactive ingredients: glycerin, dibasic sodium phosphate, metacresol, zinc oxide (zinc ion), and Water for Injection
- Hydrochloric acid and/or sodium hydroxide may be added to adjust pH
What is Admelog® (insulin lispro injection) 100 Units/mL?
Prescription Admelog is a fast-acting human insulin used to improve blood sugar control in adults with Type 2 diabetes and adults and children (3 years and older) with Type 1 diabetes.
Important Safety Information
Important Safety Information
Do not use Admelog if you have low blood sugar or if you are allergic to insulin lispro or any of the other ingredients in Admelog.
Do not share needles, insulin pens, or syringes with others, even if the needle is changed. Do NOT reuse needles. You may give other people a serious infection, or get a serious infection from them.
Change (rotate) your injection sites within the area you chose with each dose to reduce your risk of getting lipodystrophy (pitted or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites. Do not use the same spot for each injection or inject where the skin is pitted, thickened, lumpy, tender, bruised, scaly, hard, scarred or damaged.
Before starting Admelog, tell your doctor about all your medical conditions, including if you have liver or kidney problems, if you are pregnant or planning to become pregnant or if you are breastfeeding or planning to breastfeed.
Heart failure can occur if you are taking insulin together with pills called TZDs (thiazolidinediones), even if you have never had heart failure or other heart problems. If you have heart failure, it may get worse while you take TZDs with Admelog. Your treatment with TZDs and Admelog may need to be changed or stopped by your doctor if you have new or worsening heart failure. Tell your doctor if you have any new or worsening symptoms, including:
- Shortness of breath
- Sudden weight gain
- Swelling of your ankles or feet
Tell your doctor about all the medications you take, including OTC medicines, vitamins, supplements, and herbal supplements.
When used as a mealtime insulin, Admelog should be taken within 15 minutes before a meal or immediately after a meal. Test your blood sugar levels as directed by your doctor when using Admelog. Do not change your dose or type of insulin without talking to your doctor. Any change of insulin should be made cautiously and only under medical supervision.
Check your insulin label to verify that you have the correct insulin before each injection. Admelog must only be used if the solution is clear and colorless with no particles visible.
While using Admelog, do not drive or operate heavy machinery until you know how Admelog affects you. Do not drink alcohol or use medicines that contain alcohol.
Admelog may cause serious side effects that can lead to death, including: - Low blood sugar (hypoglycemia). Signs and symptoms of low blood sugar may include dizziness or light-headedness, sweating, confusion, headache, blurred vision, slurred speech, shakiness, fast heartbeat, anxiety, irritability or mood changes, hunger.
- Serious allergic reactions. Get medical help right away if you have: a rash over your whole body, trouble breathing, fast heartbeat, feeling faint, or sweating.
- Low potassium in your blood (hypokalemia). Your doctor should monitor you for low blood potassium levels.
- Heart failure. Your doctor should monitor you closely while you are taking TZDs with Admelog.
Get emergency medical help if you have:
- Trouble breathing, shortness of breath, fast heartbeat, swelling of your face, tongue, or throat, sweating, extreme drowsiness, dizziness, confusion.
Admelog may have additional side effects including injection site reactions which may include change in fat tissue, skin thickening, redness, swelling and itching.
Important Safety Information for Admelog® (insulin lispro injection) SoloStar®
Admelog SoloStar is a disposable single-patient-use prefilled insulin pen. Talk to your doctor about proper injection technique and follow instructions in the Instruction Leaflet that comes with the pen.
Important Safety Information for Admelog (insulin lispro injection) when used in a pump
Do not mix or dilute Admelog when used in a pump.
Failure of your insulin pump or infusion set or degradation of the insulin in the pump can cause hyperglycemia and ketoacidosis. Always carry an alternate form of insulin administration in the case of pump failure.
Click here for Full Prescribing Information for Admelog.
Click here for information on Sharps Medical Waste Disposal.
Click here to learn more about Sanofi's commitment to fighting counterfeit drugs.
These are not all the possible side effects of Admelog. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-10881-800-FDA-1088.
The health information contained herein is provided for general educational purposes only. Your healthcare professional is the single best source of information regarding your health. Please consult your healthcare professional if you have any questions about your health or treatment.
*Eligibility Restrictions & Offer Terms:
Sanofi Insulins Co-pay Savings Program: This savings program is not insurance. For a complete list of participating brands, products, and National Drug Codes (NDCs) Click Here. This offer is not valid for prescriptions covered by or submitted for reimbursement, in whole or in part, under Medicare, Medicaid, VA, DOD, TRICARE, similar federal or state programs, including any state pharmaceutical programs. If you have an Affordable Care (Health Care Exchange) plan, you may still be qualified to receive and use this savings card. Please note: the Federal Employees Health Benefits (FEHB) Program is not a federal or state government health care program for purposes of the savings program. Void where prohibited by law. For the duration of the program, eligible commercially insured patients pay no more than $35 per 30-day supply, up to 10 packs per fill; Offer valid for one fill every 30 days. Savings may vary depending on patients’ out-of-pocket costs. The Sanofi Insulins Co-pay Savings Program applies to the cost of medication. There are other relevant costs associated with overall treatment. Sanofi reserves the right to rescind, revoke, terminate, or amend this offer, eligibility, and terms of use at any time without notice. Upon registration, patients will receive all program details. For questions regarding your eligibility or benefits, or if you wish to discontinue your participation, call the Sanofi Insulins Co-pay Savings Program at (866) 255-8661(866) 255-8661 (9:00 am-7:00 pm EST, Monday-Friday).
Insulins Valyou Savings Program: This savings program is not insurance. For a complete list of participating brands, products, and National Drug Codes (NDCs) Click Here. This offer is only valid for those who are uninsured or those who are insured by a prescription plan but are not using such insurance and will be paying the full retail price for the medication. Void where prohibited by law. The Savings Program applies to the cost of medication. There are other relevant costs associated with overall treatment. You may not submit claims for reimbursement to any third-party payor, including any government healthcare plan (e.g., Medicare, Medicaid, DOD, VA, TRICARE) or similar federal or state programs for Sanofi Insulin prescriptions when using this Program. You may not seek to have your out-of-pocket costs or the full retail price of the Sanofi Insulin count toward your deductible, true-out-of-pocket (TrOOP), maximum out-of-pocket (MOOP), or any other out-of-pocket caps associated with any insurance coverage. Sanofi reserves the right to rescind, revoke, terminate, or amend this offer, eligibility, and terms of use at any time without notice. Upon registration, patients will receive all program details. For questions regarding your eligibility or benefits, or if you wish to discontinue your participation, call the Insulins Valyou Savings Program at (833) 813-0190(833) 813-0190 (9:00 am-7:00 pm EST, Monday-Friday).
Important Safety Information
Do not use Admelog if you have low blood sugar or if you are allergic to insulin lispro or any of the other ingredients in Admelog.
Do not share needles, insulin pens, or syringes with others, even if the needle is changed. Do NOT reuse needles. You may give other people a serious infection, or get a serious infection from them.
Change (rotate) your injection sites within the area you chose with each dose to reduce your risk of getting lipodystrophy (pitted or thickened skin) and localized cutaneous amyloidosis (skin with lumps) at the injection sites. Do not use the same spot for each injection or inject where the skin is pitted, thickened, lumpy, tender, bruised, scaly, hard, scarred or damaged.
Before starting Admelog, tell your doctor about all your medical conditions, including if you have liver or kidney problems, if you are pregnant or planning to become pregnant or if you are breastfeeding or planning to breastfeed.
Heart failure can occur if you are taking insulin together with pills called TZDs (thiazolidinediones), even if you have never had heart failure or other heart problems. If you have heart failure, it may get worse while you take TZDs with Admelog. Your treatment with TZDs and Admelog may need to be changed or stopped by your doctor if you have new or worsening heart failure. Tell your doctor if you have any new or worsening symptoms, including:
- Shortness of breath
- Sudden weight gain
- Swelling of your ankles or feet
Tell your doctor about all the medications you take, including OTC medicines, vitamins, supplements, and herbal supplements.
When used as a mealtime insulin, Admelog should be taken within 15 minutes before a meal or immediately after a meal. Test your blood sugar levels as directed by your doctor when using Admelog. Do not change your dose or type of insulin without talking to your doctor. Any change of insulin should be made cautiously and only under medical supervision.
Check your insulin label to verify that you have the correct insulin before each injection. Admelog must only be used if the solution is clear and colorless with no particles visible.
While using Admelog, do not drive or operate heavy machinery until you know how Admelog affects you. Do not drink alcohol or use medicines that contain alcohol.
Admelog may cause serious side effects that can lead to death, including: - Low blood sugar (hypoglycemia). Signs and symptoms of low blood sugar may include dizziness or light-headedness, sweating, confusion, headache, blurred vision, slurred speech, shakiness, fast heartbeat, anxiety, irritability or mood changes, hunger.
- Serious allergic reactions. Get medical help right away if you have: a rash over your whole body, trouble breathing, fast heartbeat, feeling faint, or sweating.
- Low potassium in your blood (hypokalemia). Your doctor should monitor you for low blood potassium levels.
- Heart failure. Your doctor should monitor you closely while you are taking TZDs with Admelog.
Get emergency medical help if you have:
- Trouble breathing, shortness of breath, fast heartbeat, swelling of your face, tongue, or throat, sweating, extreme drowsiness, dizziness, confusion.
Admelog may have additional side effects including injection site reactions which may include change in fat tissue, skin thickening, redness, swelling and itching.
Important Safety Information for Admelog® (insulin lispro injection) SoloStar®
Admelog SoloStar is a disposable single-patient-use prefilled insulin pen. Talk to your doctor about proper injection technique and follow instructions in the Instruction Leaflet that comes with the pen.
Important Safety Information for Admelog (insulin lispro injection) when used in a pump
Do not mix or dilute Admelog when used in a pump.
Failure of your insulin pump or infusion set or degradation of the insulin in the pump can cause hyperglycemia and ketoacidosis. Always carry an alternate form of insulin administration in the case of pump failure.
Click here for Full Prescribing Information for Admelog.
Click here for information on Sharps Medical Waste Disposal.
Click here to learn more about Sanofi's commitment to fighting counterfeit drugs.
These are not all the possible side effects of Admelog. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-10881-800-FDA-1088.
The health information contained herein is provided for general educational purposes only. Your healthcare professional is the single best source of information regarding your health. Please consult your healthcare professional if you have any questions about your health or treatment.
*Eligibility Restrictions & Offer Terms:
Sanofi Insulins Co-pay Savings Program: This savings program is not insurance. For a complete list of participating brands, products, and National Drug Codes (NDCs) Click Here. This offer is not valid for prescriptions covered by or submitted for reimbursement, in whole or in part, under Medicare, Medicaid, VA, DOD, TRICARE, similar federal or state programs, including any state pharmaceutical programs. If you have an Affordable Care (Health Care Exchange) plan, you may still be qualified to receive and use this savings card. Please note: the Federal Employees Health Benefits (FEHB) Program is not a federal or state government health care program for purposes of the savings program. Void where prohibited by law. For the duration of the program, eligible commercially insured patients pay no more than $35 per 30-day supply, up to 10 packs per fill; Offer valid for one fill every 30 days. Savings may vary depending on patients’ out-of-pocket costs. The Sanofi Insulins Co-pay Savings Program applies to the cost of medication. There are other relevant costs associated with overall treatment. Sanofi reserves the right to rescind, revoke, terminate, or amend this offer, eligibility, and terms of use at any time without notice. Upon registration, patients will receive all program details. For questions regarding your eligibility or benefits, or if you wish to discontinue your participation, call the Sanofi Insulins Co-pay Savings Program at (866) 255-8661(866) 255-8661 (9:00 am-7:00 pm EST, Monday-Friday).
Insulins Valyou Savings Program: This savings program is not insurance. For a complete list of participating brands, products, and National Drug Codes (NDCs) Click Here. This offer is only valid for those who are uninsured or those who are insured by a prescription plan but are not using such insurance and will be paying the full retail price for the medication. Void where prohibited by law. The Savings Program applies to the cost of medication. There are other relevant costs associated with overall treatment. You may not submit claims for reimbursement to any third-party payor, including any government healthcare plan (e.g., Medicare, Medicaid, DOD, VA, TRICARE) or similar federal or state programs for Sanofi Insulin prescriptions when using this Program. You may not seek to have your out-of-pocket costs or the full retail price of the Sanofi Insulin count toward your deductible, true-out-of-pocket (TrOOP), maximum out-of-pocket (MOOP), or any other out-of-pocket caps associated with any insurance coverage. Sanofi reserves the right to rescind, revoke, terminate, or amend this offer, eligibility, and terms of use at any time without notice. Upon registration, patients will receive all program details. For questions regarding your eligibility or benefits, or if you wish to discontinue your participation, call the Insulins Valyou Savings Program at (833) 813-0190(833) 813-0190 (9:00 am-7:00 pm EST, Monday-Friday).



